Ca2+ release-activated Ca2+ (CRAC) channel comprising STIM and ORAI proteins is a prototypical example of store-operated Ca2+ entry (SOCE) and serves as an indispensable high-ly-selective Ca2+ entry route in both excitable and non-excitable cells. SOCE is initiated by Ca2+ store depletion within the lu-men of the endoplasmic reticulum (ER) and subsequent multi-merization of the STIM1 ER-luminal domain. Activated STIM1 molecules accumulate at ER-PM junctions to engage and directly gate ORAI1 channels, permitting Ca2+ flux into cells. Aberrant STIM-ORAI signaling has been linked to several hu-man diseases, including immunodeficiency, autoim-munoinflammatory disorders, cardiac hypertrophy, cancer me-tastasis, and the Stormorken syndrome. Stormorken syn-drome is believed to arise from gain-of-function mutations in either ORAI1 or STIM1 (e.g., R304W) that lead to constitutive Ca2+ entry. Therefore, CRAC channel has been actively pur-sued as a promising therapeutic target.
Recently, in collaboration with teams at Texas A&M University and Beijing Normal University, Professor Li Minyong's group at the School of Pharmaceutical Sciences published a series of photoswitchable CRAC channel modulators named as piCRACs through bioisosteric replacement in heterocyclic N-aryl benzamide derived SOCE inhibitors, in Journal of the American Chemical Society (J. Am. Chem. Soc.2020,doi: 10.1021/jacs.0c02949).
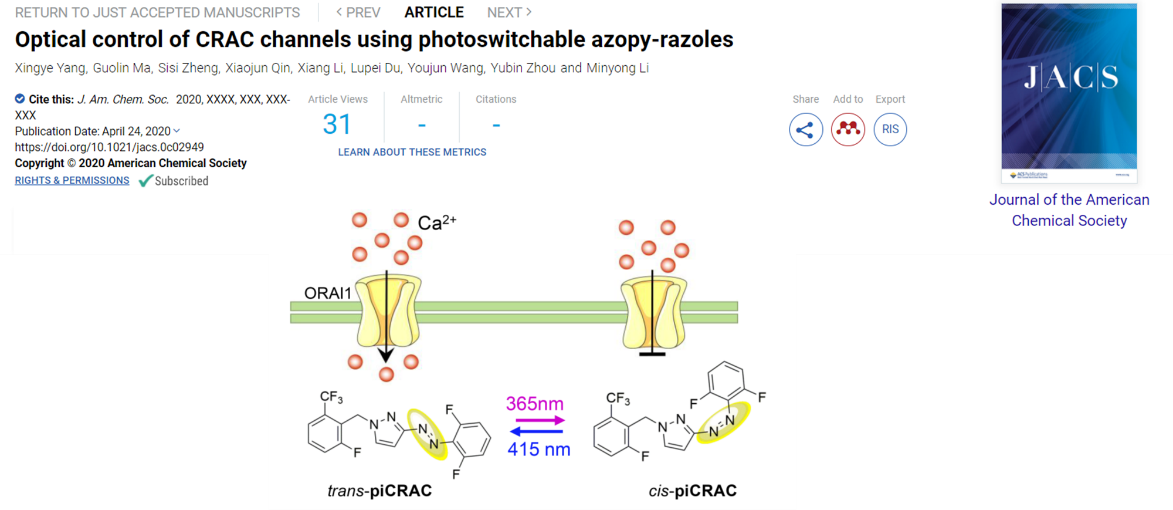
Among these molecules, PiCRAC-1 permits light-inducible modulation of both CRAC channels and Ca2+-dependent physiological processes in cellulo with appropriate spatiotemporal precision. Most excitingly, piCRAC-1 has been successfully demonstrated as a potential therapeutic agent to optically intervene in pathological conditions associated with dysregulated Ca2+ signaling, such as the Stormorken syndrome arising from over-activating mutations of CRAC channels in zebrafish model.
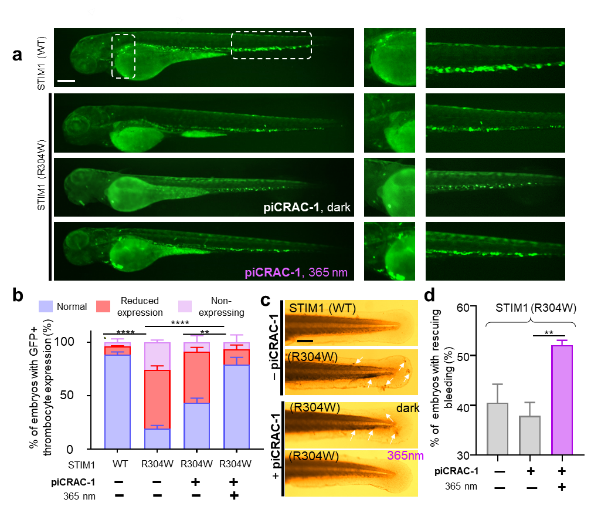
PiCRAC-1 takes action in its cis-configuration, making it a new addition to the cis-on photopharmacology toolbox. This chemical biology toolkit is anticipated to not only facilitate the study of structure-function relations of Ca2+ channels, but also has the potential to interrogate a myriad of Ca2+-regulated signaling events and human disorders associated with imbalanced Ca2+ homeostasis.
Professor Li Minyong's group has been working on visualization analysis-based drug discovery. IN recent years, his team has published a series of papers on various prestigious journals such as Chem. Soc. Rev., Anal. Chem., J. Med. Chem., Chem. Commun., etc., which have been highlighted by various journals and internet media. The current work is supported by the National Natural Science Foundation of China, the Distinguished Young Scholars Program of Shandong University, the Taishan Scholar Program, the Shandong Natural Science Foundation, and the Key Research and Development Project of Shandong Province.
Written by:Li Linwei
Edited by: Che Huiqing