Recently, researchers from the State Key Laboratory of Crystal Materials, Institute of Marine Science and Technology and Advanced Medical Research Institute report that wireless localized electrical stimulation generated by an ultrasound-driven piezoelectric discharge can regulate macrophages polarization. The strategy provides an effective anti-tumor method and has opened up a new way for tumor immunotherapy. Relevant paper entitled ‘Wireless Localized Electrical Stimulation Generated by an Ultrasound-Driven Piezoelectric Discharge Regulates Proinflammatory Macrophage Polarization’ has been published online in Advanced Science (2021, DOI:10.1002/advs.202100962, IF: 15.5). Kong Ying and Liu Feng in the School of Crystal Materials are co-first authors of this paper. Prof. Liu Hong and Prof. Wang Shuhua in the State Key Laboratory of Crystal Materials, and Prof. Han Lin in theInstitute of Marine Science and Technologyare co-corresponding authors.

Immune cells are an important part of the human immune system. The immune defense function of immune cells can resist the invasion of bacteria and viruses, and the immune surveillance function can remove pathological and cancerous cells in time. Immune cells are the health guard of human beings. Macrophages are one of the most important innate immune cells in vertebrate including human beings, and have different polarization states, including the proinflammatory (M1) phenotype and anti-inflammatory (M2) phenotype. Proinflammatory macrophages kill cancer cells or inhibit tumor growth, and regulating the M1 polarization of macrophages has become a very hot topic in the cell technology field. The immune process and macrophage polarization are routinely regulated by biological and chemical factors. Although much progress has been made in elucidating the mechanism underlying the regulation of the immune cell response based on pattern recognition receptors andmicroenvironmental sensors, it is still a great challenge to identify rapid and highly efficient methods that drive the M1 polarization of macrophages. Recently, there has been great interest in the regulation of immune cells by physical signals. As an important type of physical stimulation, electrical signals have aroused increasing attention in the biomedical field and can directly program cellular behavior in an electrogenic-mediated manner. However, few studies have focused on how electrical signals regulate macrophage polarization. Electrical stimulation may have an important effect on macrophage polarization through regulating the cellular microenvironment.
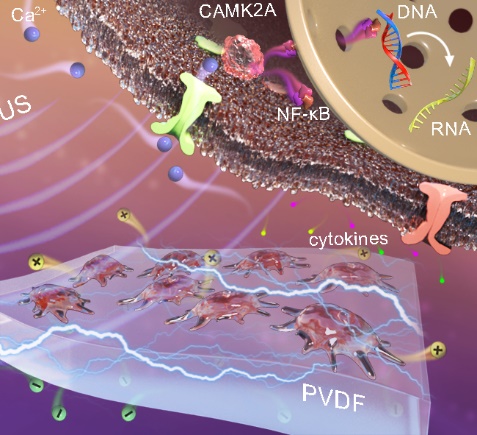
Prof. Liu Hong’s group designed a strategy to noninvasively enhance the proinflammatory response of macrophages through piezoelectric material-meditated localized electrical signals with ultrasound assistance. This strategy relies on the micro-vibration of the β-PVDF film under ultrasound treatment and the release of a localized charge via the spontaneous polarization of the crystalline phase, which significantly triggers the selective expression and secretion of proinflammatory chemoattractant factors. Based on RNA sequencing, Ca2+influx through voltage-gated channels is promoted, and a Ca2+-CAMK2A-NF-κB axis is established to induce M1 macrophage polarization. Following coculture with M1-polarized macrophages, tumor cell viability is significantly inhibited due to macrophage-secreted inflammatory factors. This study strongly supports that piezoelectric material-mediated electrical signals mainly regulate immunological responses, providing controllable noninvasive stimulus to drive the inflammatory response and suggesting the potential applications of this strategy as a tumor immunotherapy.
This work was supported by the National Key R&D Program of China, National Natural Science Foundation of China, Natural Science Foundation of Shandong Province, Major Innovation Projects in Shandong Province, the Project of “20 items of University” of Jinan, and the Young Elite Scientist Sponsorship Program by CAST (YESS). The Collaborative Innovation Center of Technology and Equipment for Biological Diagnosis and Therapy in Universities of Shandong are thanked.
Link:https://onlinelibrary.wiley.com/doi/10.1002/advs.202100962